COA PHARMACY ASSOCIATION (COPA)
A Network of
SupportToolsCommunityResourcesCare
for Oncology & Urology Pharmacies
Supporting and Advocating for Independent Medically Integrated Pharmacies and the Patients they Serve
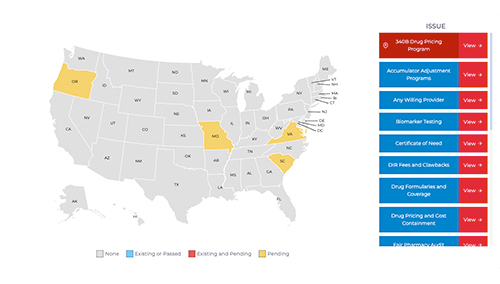
Our Most Useful Tools
COPA has current and frequently updated tools available to help elevate some of the tasks on the community oncology pharmacies.