Should All These Drugs Really Be Considered ‘Orphans’?
The authors of a new report in the American Journal of Clinical Oncology accuse pharmaceutical companies of gaming the Orphan Drug Act (ODA) to maximize profits from drugs ultimately used for large populations of patients, thus driving up medication costs for patients with cancer and diluting incentives designed to benefit patients with rare cancers.
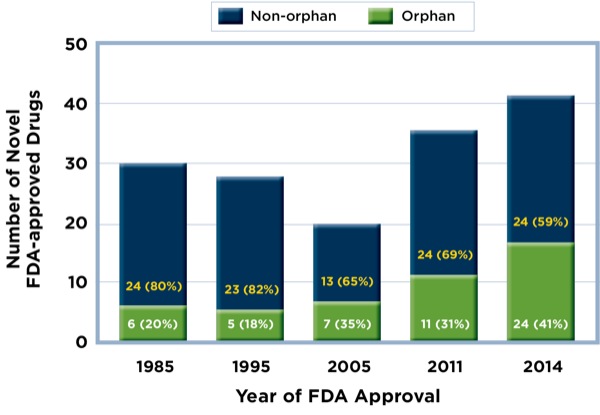
The new report presents data showing a marked increase in use of the ODA in recent years. Since the law was passed in 1983, the proportion of new agents submitted as orphan drugs and ultimately approved by the FDA has risen steadily (Figure). In 2014, 41% of all FDA-approved drugs were approved as orphan drugs.
“The ODA is responsible for numerous chemotherapeutic advancements,” acknowledged Michael G. Daniel, MD, and his colleagues from the Johns Hopkins University School of Medicine, in Baltimore, in the report published online (2015 Nov 17. [Epub ahead of print]). “However,” they added, “the ODA is also responsible for pharmaceutical firms gaining unintended market profits.”
Many drugs approved under the ODA have become mainstream. Rituximab (Rituxan, Genentech), originally approved with an orphan designation for the treatment of follicular non-Hodgkin lymphoma, is now the 12th all-time best-selling medication in the United States, generating over $3.7 billion in sales in 2014, according to the authors.
Drugs designated as orphan drugs benefit from protections and exclusivity benefits—incentives to induce companies to invest in diseases with such small patient populations that investment would be otherwise unprofitable. Dr. Daniel and his co-authors noted that ODA incentives include federal grants, a tax credit equaling 50% of the qualifying clinical trial costs, a waiver of marketing application user fees worth in excess of $2 million and seven years of marketing exclusivity.
The trend for increasing numbers of orphan drugs is particularly pronounced with new cancer therapies, for which companies stratify patient populations into smaller and smaller groups. The current system of classifying cancer medications by both the organ of origin and the molecular identity is “creating a situation wherein almost every medication can be an orphan,” Dr. Daniel and his co-authors wrote.
Continue Reading on Clinical Oncology News